Cannizzaro Response | ChemTalk
[ad_1]
Core Ideas
On this tutorial, you’ll be taught concerning the Cannizzaro response, together with its mechanism, purposes, and examples.
Matters Coated in Different Articles
Cannizzaro Response Overview
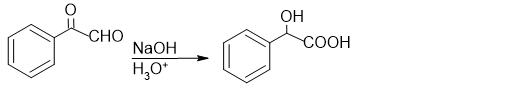
The Cannizzaro response is a redox response that entails the simultaneous oxidation and discount of two molecules of aldehyde that lack alpha hydrogen into carboxylic acid and alcohol. Found in 1853 by Italian chemist Stanislao Cannizzaro, the response received first noticed in benzaldehyde, which gave benzyl alcohol and potassium benzoate when handled with potash.
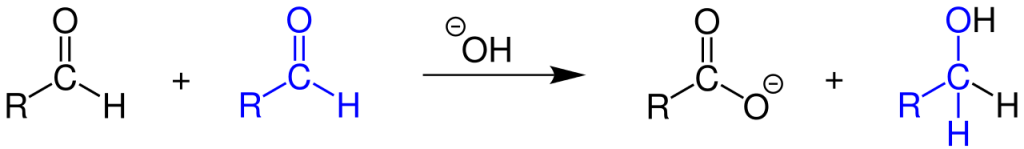
It’s is an instance of a disproportionation response as one molecule (aldehyde) undergoes simultaneous oxidation and discount. It’s a base-induced response the place the hydroxide group from the bottom act as a robust nucleophile to provoke the response.
Each of the alcohol and acids produced have an important organic significance and can be utilized within the manufacturing of meals preservatives, antiseptics and antibacterial medicine.
Cannizzaro Response Reactants
Aldehyde
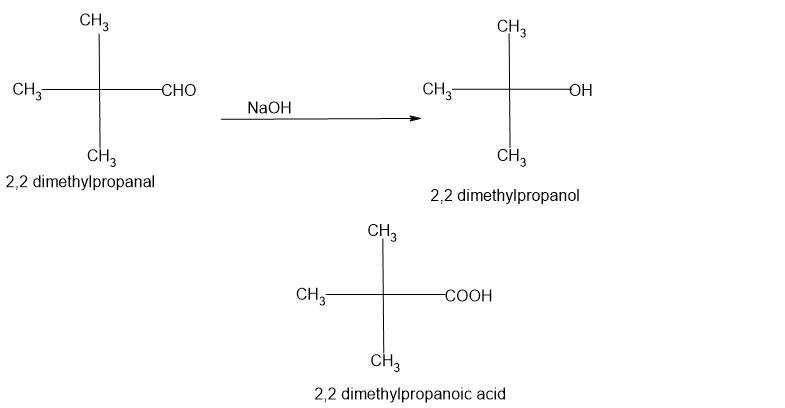
Two aldehydes molecules which lack energetic hydrogen at alpha carbon, also called non-enolizable aldehydes, act as reactant molecules. Instance: Formaldehyde, Benzaldehyde, Vanillin
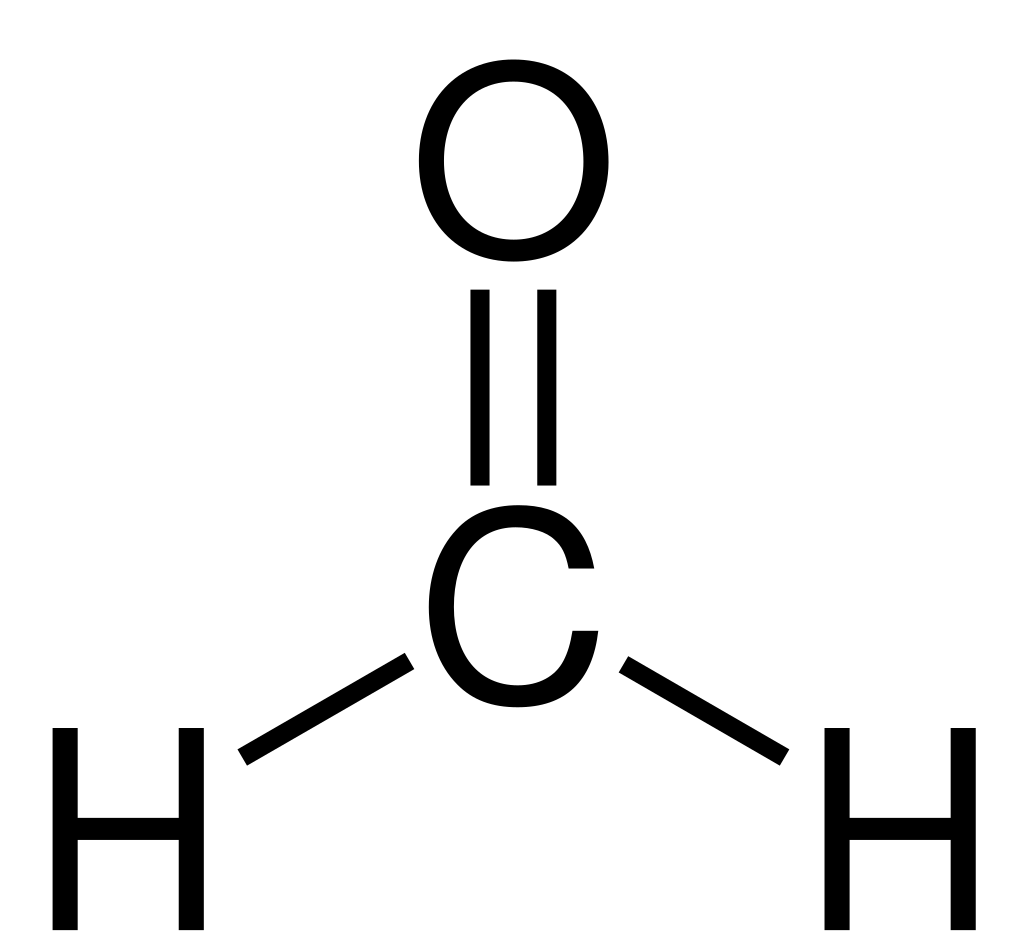
Base
Excessive focus of sturdy base like sodium hydroxide and potassium hydroxide is used. The bottom generates electron wealthy hydroxide ions which assaults the carbonyl group of aldehyde and promote a disproportionation response.
Cannizzaro Response Mechanism
1.Because of the electron-withdrawing nature of bonded oxygen atoms, the carbon within the carbonyl group possesses a partial constructive cost and turns into an energetic website for nucleophilic assault by the hydroxide ion. The incoming nucleophile assaults the carbon within the carbonyl group and displaces the electron from the pi bond onto the oxygen atom, giving it a adverse cost. This varieties a tetrahedral intermediate, from which the hydroxide ion once more abstracts hydrogen to kind an anion with two adverse expenses often known as a dianion.
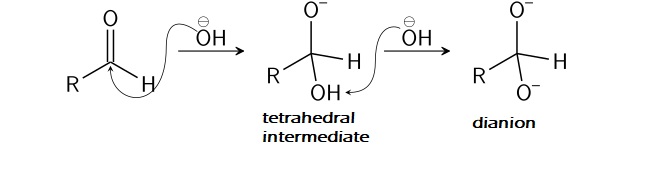
2. The negatively charged oxygen on the dianion reforms the C to O double bond, and the removing of the hydride ion as a leaving group takes place. After this, the hydride ion assaults the carbonyl group of one other molecule and converts the dianion to a carboxylate anion and the impartial aldehyde to an alkoxide anion. Thus shaped alkoxide anion is protonated by water molecule to present alcohol.
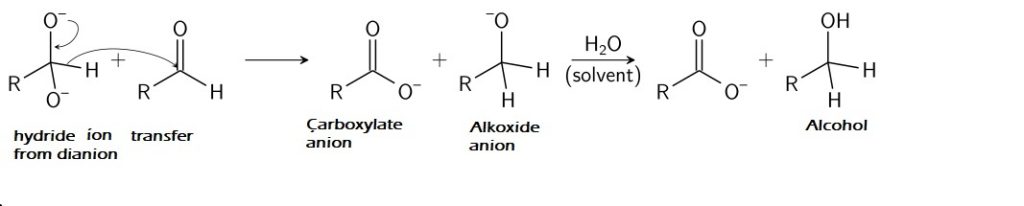
This hydride switch is a gradual step and therefore, the rate-determining step in Cannizzaro response.
Cross Cannizzaro Response
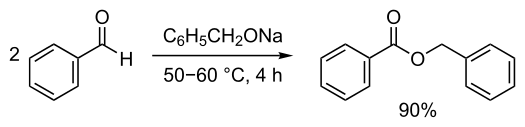
If an aldehyde with out alpha hydrogen is reacted with formaldehyde within the presence of a robust base to kind formic acid and alcohol, this response is named the cross Cannizzaro response. Right here, formaldehyde acts as a reductant and is combined with one other aldehyde molecule for the manufacturing of the required alcohol. On this response, the entire transformation of the 2 totally different reactants takes place to present the specified product.
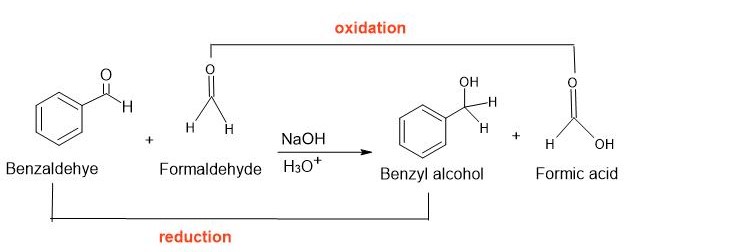
Cannizzaro Response Utility
- The mix of crossed Cannizzaro response and aldol condensation results in the manufacturing of Polyols
- A two stepped base catalyzed response involving aldol condensation and Cannizzaro response varieties trimethylolpropane. Trimethylolpropane is a precursor to alkyd resin and utilized throughout the manufacture glues, dyes, silicone merchandise, and artificial lubricants
- The benzyl alcohol generated by the next course of reveals anesthetic and antipruritic properties. Because of the antibacterial and antifungal nature of benzyl alcohol, it additionally acts as a preservative. Equally, benzoic acid can also be an essential antimicrobial preservative within the meals and drinks trade.
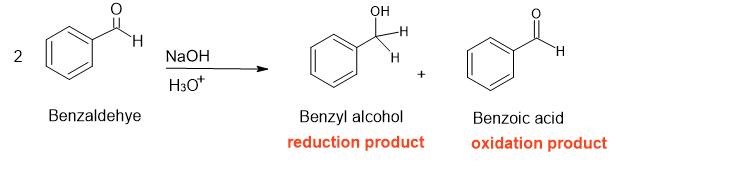
Observe issues
[ad_2]