Calorimetry | ChemTalk
[ad_1]
Core Ideas
On this article, you’ll study calorimetry and calorimeters, primarily used to calculate particular warmth capability and thermal adjustments in a bodily or chemical response. Ideas like various kinds of calorimeters and the calorimeter equation can even be coated.
Matters Coated in Different Articles
What’s Calorimetry?
Calorimetry is a subject of thermochemistry that measures the quantity of warmth concerned in a bodily or chemical response. Chemists use calorimetry to find out the quantity of warmth transferred to or from a system into its environment. From a chemical response like acid-base neutralisation to a bodily response like solidifying, calorimetry permits scientists to measure thermal adjustments inside any response.
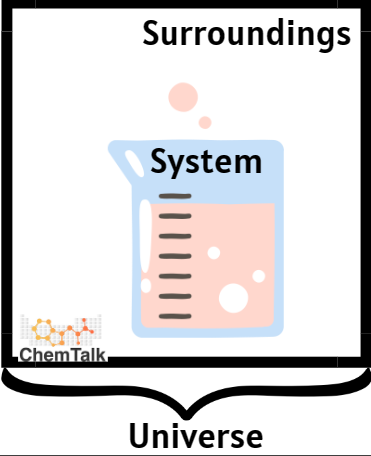
To measure warmth change, chemists first outline the response’s system vs. environment. In thermodynamics, a system is the substance/substances present process the chemical or bodily response, whereas the environment is all different matter. The environment serve to both present or take in warmth to the system. The temperature distinction between environment and system is then used to derive the quantity of warmth transferred by the response.
What’s a Calorimeter?
Essential to calorimetry, a calorimeter is any machine used to measure the quantity of warmth concerned in a surroundings-system switch. The change in temperature can be utilized with particular warmth and mass to find out the quantity of warmth concerned in a response.
A calorimeter can be used to find out the particular warmth capability of sure substances concerned within the warmth switch response.
There are numerous various kinds of calorimeters that accomplish particular capabilities and measure sure qualities of a substance. Nonetheless, most calorimeters include a metallic vessel which is well-insulated to stop further vitality from escaping into the environment.
This vessel’s solely opening is for the thermometer to measure adjustments within the response’s thermal properties. This thermometer measures important adjustments in warmth, permitting chemists to find out warmth capacities and the entire vitality used within the response.
Exothermic vs. Endothermic Reactions
An endothermic response is when the system or response features vitality from its environment. For instance, dissolving an answer of ammonium nitrate in water is an endothermic chemical course of as a result of the water loses thermal vitality (which explains how instantaneous chilly packs work)!
Then again, endothermic reactions might be defined by the equation beneath, the place q represents warmth. Discover that the system has a constructive q-value, indicating that the system gained warmth by the response. Then again, the environment has a adverse q-value as a result of the environment misplaced warmth to the system.
![]()
Exothermic reactions contain the system releasing vitality into the environment – any chemical response that produces warmth, like an instantaneous sizzling pack, is an exothermic response. In these reactions, the system loses vitality whereas the environment acquire vitality, as represented by the equation beneath.
![]()
Furthermore, the quantity of vitality transferred from methods to environment should all the time equal one another. Subsequently, this elementary phenomenon abides by the First Regulation of Thermodynamics – which states that vitality can by no means be destroyed or created in a response.
Calorimetry Equation
When analysing a warmth switch response, chemists use the calorimetry equation relating warmth launched within the response to the substance’s mass, change in temperature, and particular warmth capability.
Within the equations, q is the quantity of warmth measured in joules. m is mass measured in grams.
![]()
as change in temperature measured in Celsius, or the preliminary temperature subtracted from the ultimate temperature. c is the particular warmth capability of a substance, or the quantity of joules wanted to lift the temperature by 1 diploma Celsius.
When supplied with both of those values, one can simply calculate another worth within the equation and decide what signal the q-value is (indicating whether or not the response is exothermic or endothermic).
![]()
Sorts of Calorimetry
Furthermore, calorimetry falls underneath three major classes, based mostly on which components stay fixed: fixed stress calorimetry, fixed quantity calorimetry and differential scanning calorimetry.
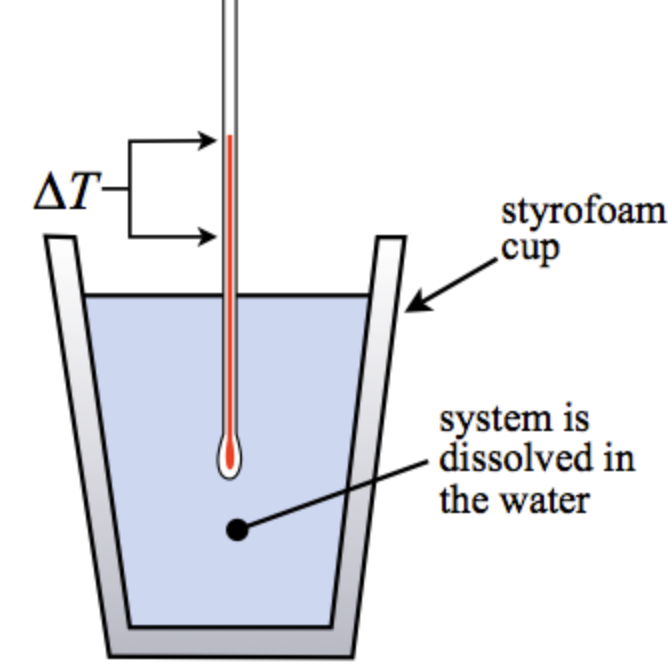
- Fixed Strain Calorimetry
By protecting exterior vs. inside stress constant, this calorimetry isolates the methods from shedding or gaining warmth from environment. Chemistsuse a espresso cup-like machine in this kind of calorimetry, which merely insulates the system.
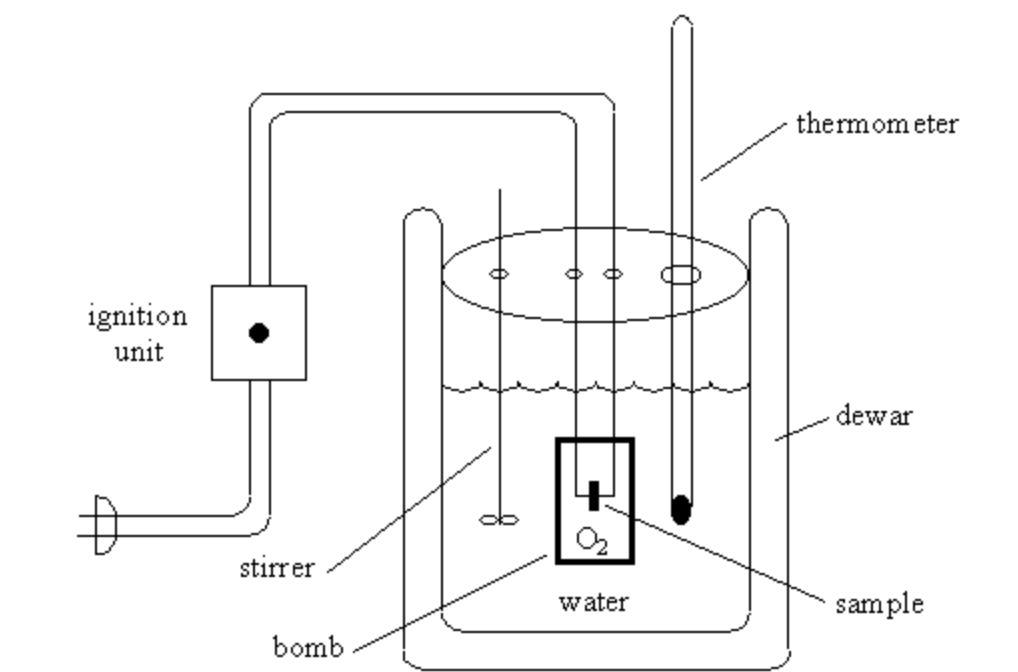
2. Fixed Quantity Calorimetry
Much like its title, fixed quantity calorimetry goals to keep up a continuing quantity and resist massive quantities of stress. This results in larger accuracy when calculating warmth switch and this calorimeter is named a “bomb calorimeter,” a tool that relates warmth of response and particular warmth capability.
3. Differential Scanning Calorimetry
Together with each a pattern and a reference chamber, differential scanning calorimetry compares particular warmth capacities of the identical solvent. The pattern and reference chambers each include the identical volumes of solvent, whereas the pattern chamber additionally comprises the substance of curiosity. Any adjustments within the warmth of response all through the response can thus be attributed to the substance, which is impacting the calculated warmth capability.
Calorimetry Observe Issues
- When 1g of glucose is combusted in a bomb calorimeter, the temperature of the calorimeter will increase by 2 levels C. The warmth capability of the calorimeter and its contents is 9.90 kJ/levels C. What’s the q-value for this combustion?
- What number of mL of water at 23 °C with a density of 1.00 g/mL should you combine with 180 mL of espresso at 95 °C in order that the ensuing mixture could have a temperature of 60 °C? Assume that espresso and water have the identical particular warmth (4.184 J/g °C).
- You place a 45-g aluminium spoon (particular warmth 0.88 J/g °C) at 24 °C in 180 mL (180 g) of espresso at 85 °C. The temperature of the 2 change into equal.
- The temperature of the cooling water because it leaves the recent engine of an car is 240 °F. After it passes by the radiator it has a temperature of 175 °F. Calculate the quantity of warmth transferred from the engine to the environment by one gallon of water with a selected warmth of 4.184 J/g °C.
- How a lot will the temperature of a cup (180 g) of espresso at 95 °C lower when a forty five g silver spoon (particular warmth 0.24 J/g °C) at 25 °C enters the espresso and the 2 attain the identical temperature? Assume that the espresso has the identical density and particular warmth as water.
Calorimetry Observe Drawback Options
- q = 19.8 kJ
- 170 mL
- 81.95 °C
- 570 kJ
- 1 diploma
[ad_2]