What Are Colloids? | ChemTalk
[ad_1]
Core Ideas
On this article, you’ll study colloids – one of many main sorts of mixtures. Past defining colloid mixtures, this text will cowl distinctive colloidal properties, establish a colloid combination, and functions of colloids in on a regular basis life. Be taught extra about a necessary combination to lifeforms – colloids!
Subjects Lined in Different Articles
Colloids
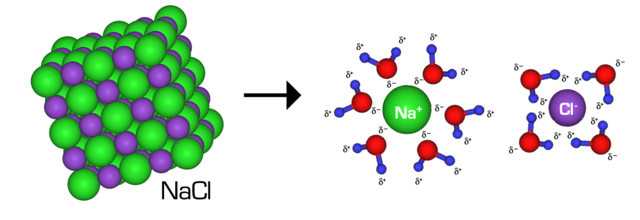
A colloid, or a colloidal resolution, is a mix consisting of molecules or particles dispersed in resolution. Not like the opposite two main forms of combination, options and suspensions, colloids comprise particles which are evenly distributed all through the answer. Furthermore, the particles suspended/dispersed belong to a unique substance than the answer they’re suspended in. As a result of colloids include particles dispersed in one other substance, the particles are known as the dispersed section whereas the substance the particles are dispersed in are known as the continual section.

These particles are microscopic and insoluble, and their sizes vary from 1 to 100 nanometers. The combination is a suspension when the particles are bigger than this vary. If the particles are smaller than this vary, the combination is an answer.
Colloidal Properties
Chemists study varied properties to find out whether or not a mix may be categorised as a colloid.
- The dispersed particles ought to by no means settle on the backside of resolution and should stay suspended in resolution.
- Centrifugation can separate colloid particles from the solvent.
- Filtration strategies can not separate colloid particles, as a result of their small measurement.
- All colloids are heterogenous mixtures, as colloid options consists of various substances dispersed in one another.
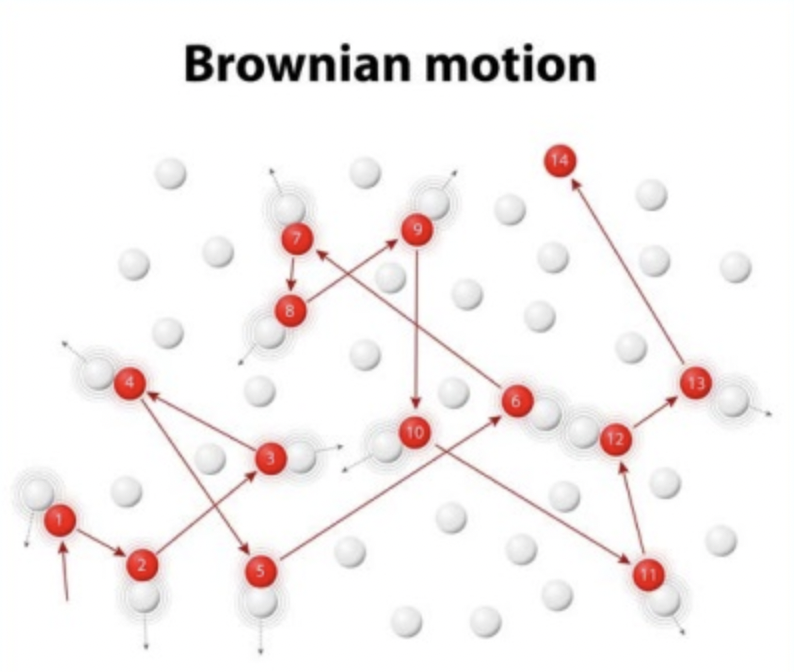
- Colloidal particles show Brownian Motion – the phenomena of random particle actions in resolution.
Tyndall Impact

Along with Brownian Movement and heterogeneity, one main property of colloids is displaying the Tyndall Impact. When somebody shines a beam of sunshine by a colloidal resolution, the sunshine scatters and displays in all instructions. For instance, consider shining a flashlight by mud or fog – that very same phenomenon happens with a real colloid. In distinction, shining mild by an answer will move cleanly by, and never refract in several instructions. The suspended particles attribute of colloids create the Tyndall Impact, and plenty of chemists will check whether or not a mix is a colloid by shining mild by it.
Colloid Preparation
When making a colloid combination, there are all kinds of preparation strategies for colloids. Some main strategies embrace chemical response, peptization, mechanical dispersion, and electrical dispersion.
- Getting ready colloids by way of chemical response can contain double alternative, oxidation, discount and hydrolysis. The merchandise of those reactions combination to type colloids. Some examples of chemical reactions that type colloid options are under.
![]()
![]()
![]()
- Peptization is when an electrolyte (or a peptizing agent) is slowly added in small increments to a precipitate. The precipitate then absorbs the electrolyte ions and turns into a colloid. Some peptide examples embrace gelatin or glucose.
- Mechanical dispersion – solutes floor into colloidal particle measurement after which blended right into a dispersion medium. Through the use of mechanical grinders at excessive speeds and revolutions, the particles forcefully disperses into colloidal suspension. This industrial methodology is used within the manufacturing of paint, black ink, and extra.
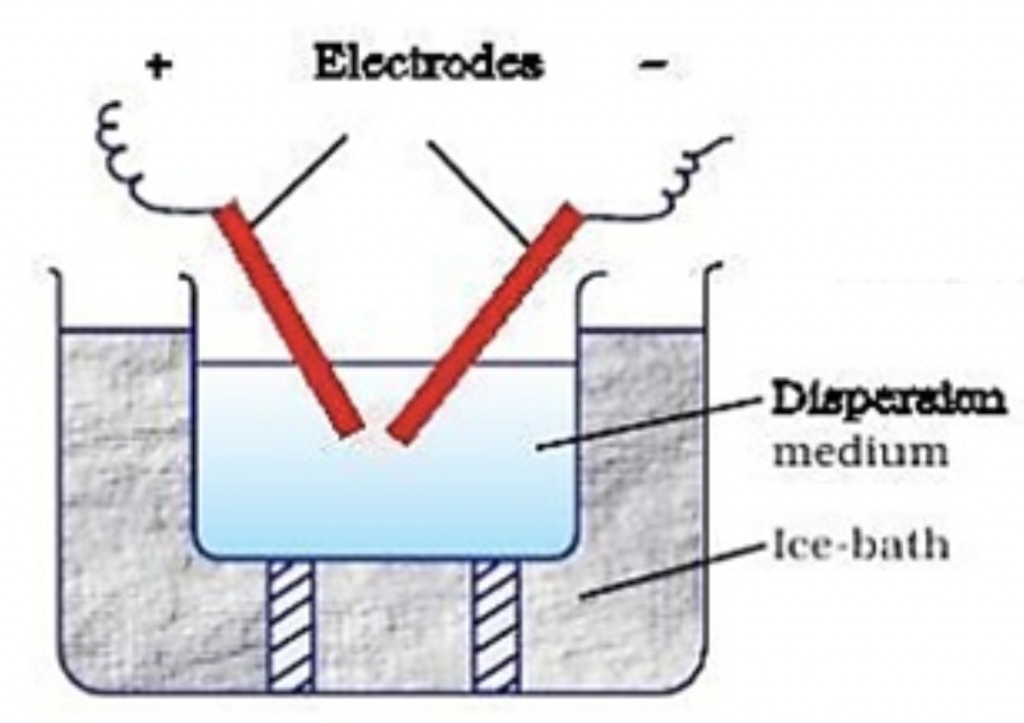
- Electrical dispersion – when {an electrical} present runs between two metallic electrodes immersed in a dispersion resolution. From the warmth generated with this methodology, a colloidal resolution varieties. Stabilisation processes primarily use this methodology.
Totally different Kinds of Colloids
Along with preparation methodology, colloids are sometimes categorised into 6 classes primarily based on their state of matter: aerosol, strong resolution, foam, gels, hydrocolloids and emulsion.
- Stable resolution – a colloidal suspension with strong particles in liquid.
- Aerosol – small strong or liquid particles dispersed in a fuel.
- Foam – fuel particles dispersed in a strong or liquid resolution.
- Gels – liquid particles dispersed inside a strong.
- Hydrocolloids – any colloid the place the dispersion medium is water.
- Emulsions – a colloid consisting of liquid particles dispersed in liquid resolution.
Examples of Colloids
Whether or not you’ve heard of them or not, colloids are throughout us in all shapes and varieties! Whether or not it’s the gray fog you get up to within the morning or the milk you drink for breakfast, colloidal options are important to every day life, in addition to the medical/biochemistry subject. Due to this fact, some examples of colloids embrace paint, automotive exhaust, lava, cheese, and all kinds of starches. Inside medical functions, colloids are essential as main bodily fluids are sometimes colloids, akin to bile or blood.


Colloids Follow Issues
Downside 1
What sort of colloid is a mix containing strong particles dispersed in a fuel?
Downside 2
What phenomenon happens if you shine mild by a colloid combination?
Downside 3
Which colloid preparation methodology entails operating {an electrical} present between two electrodes?
Downside 4
Are colloids homogenous or heterogenous mixtures?
Downside 5
What are the particles in a colloid known as?
Colloids Follow Downside Options
- Aerosols
- The Tyndall Impact
- Electrical Dispersion
- Heterogenous Mixtures
- The Dispersed Section
[ad_2]